The Art of Scaling: From Lab to Production Without Losing Your Edge
In our previous article, we established a powerful truth: sustainability is not a cost, but a direct driver of profitability. The circular economy, process efficiency, and new revenue from waste streams are creating immense value for forward-thinking chemical companies. But a brilliant, sustainable innovation in a laboratory flask is one thing; producing it consistently, safely, and profitably at a multi-ton scale is another challenge entirely.
This is the “valley of death” for chemical innovation, where countless promising ideas fail. The journey from the lab bench to a full-scale production facility is fraught with technical, financial, and logistical hurdles. As industry leaders gather for Chemspec Europe, the sessions on scaling up continuous-flow therapeutics and advanced manufacturing are not just academic discussions- they are about conquering this valley.
Successfully scaling an innovation is an art form that blends scientific rigor with engineering pragmatism. It requires a fundamental shift in mindset and a holistic approach that considers every aspect of the production process.
The Mindset Shift: From Discovery to Delivery
The skills and mindset that lead to a breakthrough in the lab are often different from those required to commercialize it. The transition from discovery to delivery involves moving from a focus on possibility to a focus on reliability.
| Aspect | Lab (Discovery) | Plant (Delivery) |
|---|---|---|
| Goal | Prove a concept works | Produce consistently at target cost & quality |
| Scale | Grams to kilograms | Tons to kilotons |
| Mindset | “What if?” | “What could go wrong?” |
| Primary Driver | Novelty and performance | Safety, reliability, and economics |
| Key Metric | Yield and purity | Overall Equipment Effectiveness (OEE) & ROI |
This shift is where many organizations falter. The R&D team that created the innovation must work hand-in-hand with process engineers, supply chain experts, and quality assurance teams from the very beginning.
The Three Pillars of Successful Scaling
Conquering the valley of death depends on mastering three critical pillars:
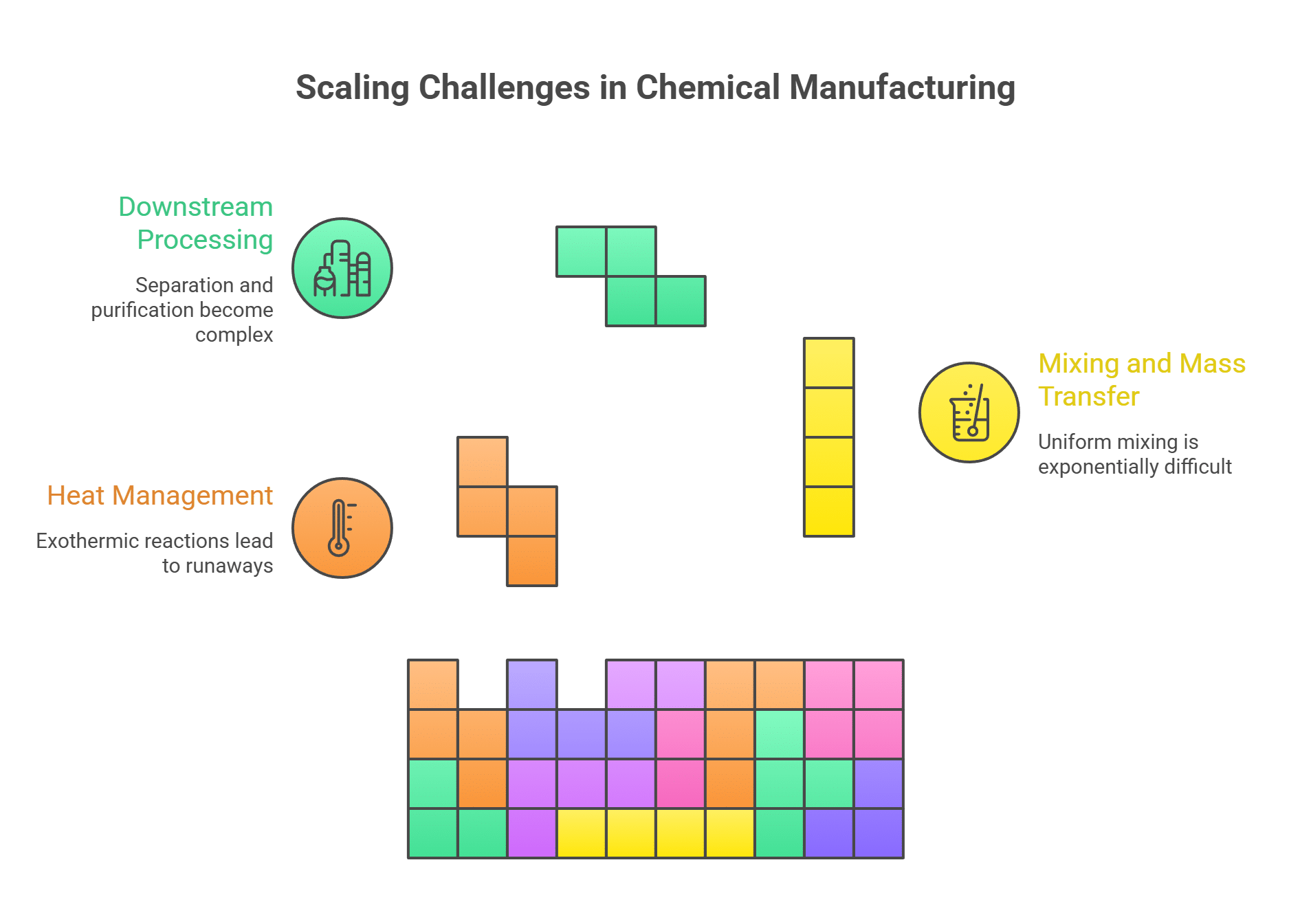
1. Robust Process Engineering
A reaction that works perfectly in a 1-liter glass flask can behave unpredictably in a 10,000-liter stainless steel reactor. The principles of thermodynamics, heat transfer, and mass transfer that are negligible at the lab scale become dominant at the industrial scale.
Key Challenges:
- Heat Management: Exothermic reactions that are easily cooled in the lab can lead to dangerous thermal runaways in a large reactor.
- Mixing and Mass Transfer: Achieving uniform mixing and reaction rates is exponentially more difficult at scale.
- Downstream Processing: Separating and purifying the final product can become the most complex and expensive part of the entire process.
The Solution: Design for Scale Successful scaling begins in the lab. Process chemists and engineers must work together to develop a process that is not just elegant, but also robust and scalable. This includes choosing reagents that are readily available at industrial scale, designing reaction conditions that are safe and controllable, and developing an efficient purification strategy from day one.
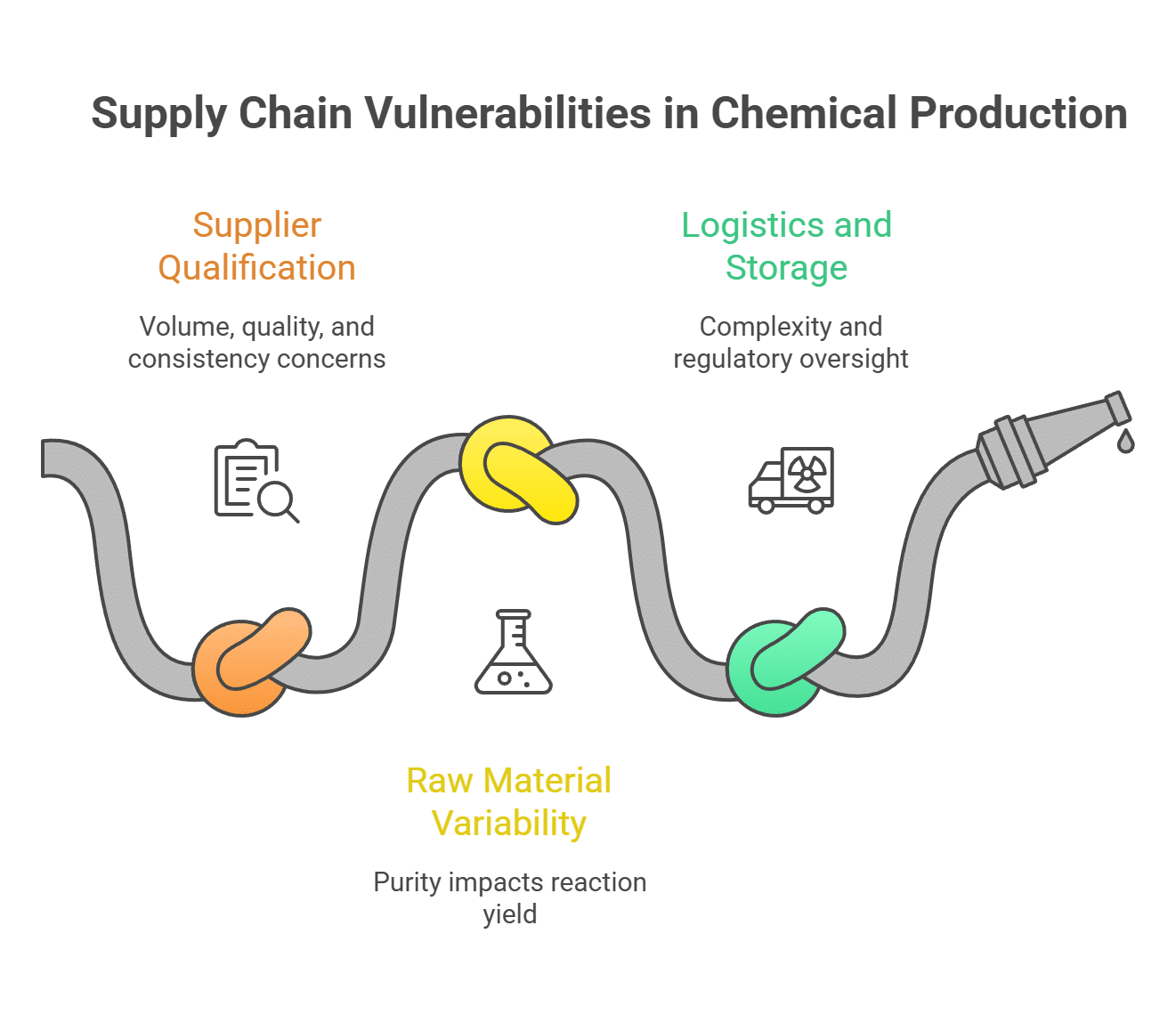
2. A Resilient and Scalable Supply Chain
The ultra-pure, lab-grade solvent you used to develop your process may be unavailable or prohibitively expensive at the volumes you need for full-scale production. Your entire process is built on the assumption of consistent, high-quality raw materials, and your supply chain is where that assumption is tested.
Key Challenges:
- Supplier Qualification: Can your chosen suppliers provide the required volume, quality, and consistency? What is their own operational resilience?
- Raw Material Variability: Even small variations in the purity of a raw material can have a significant impact on reaction yield and final product quality.
- Logistics and Storage: Sourcing, storing, and handling industrial quantities of chemicals introduces a new layer of complexity and regulatory oversight.
The Solution: A Diversified and Agile Sourcing Strategy A scalable supply chain is built on diversity and flexibility. This means having multiple qualified suppliers for critical raw materials and building relationships with partners who can provide agility. This is where a strategic approach to chemical sourcing becomes a competitive advantage. Furthermore, working with surplus chemical suppliers can provide a valuable source of alternative materials and help mitigate the risk of single-supplier disruptions.
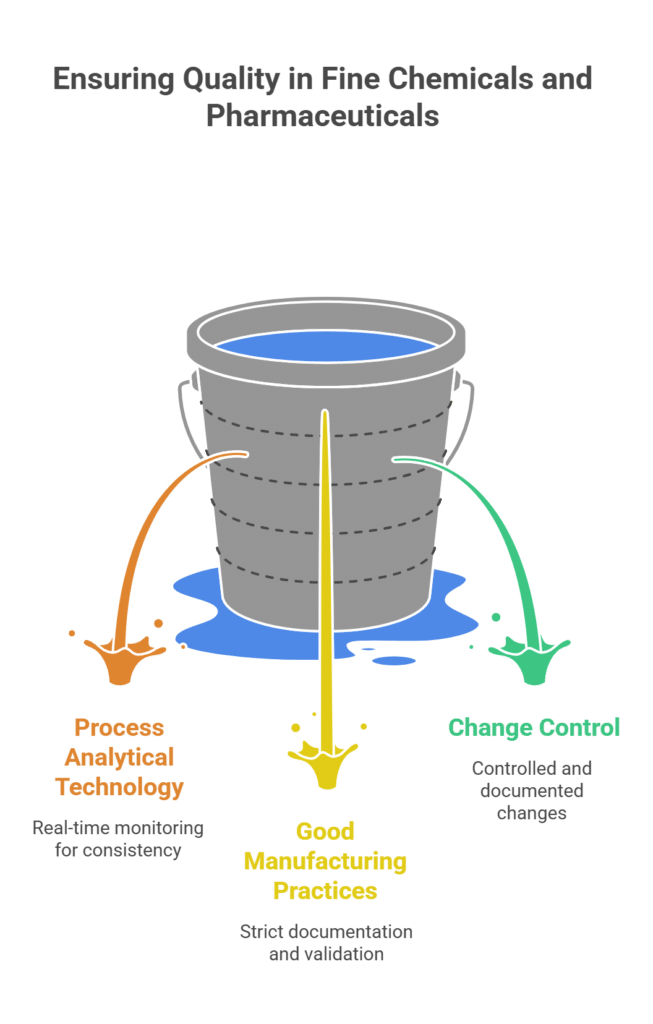
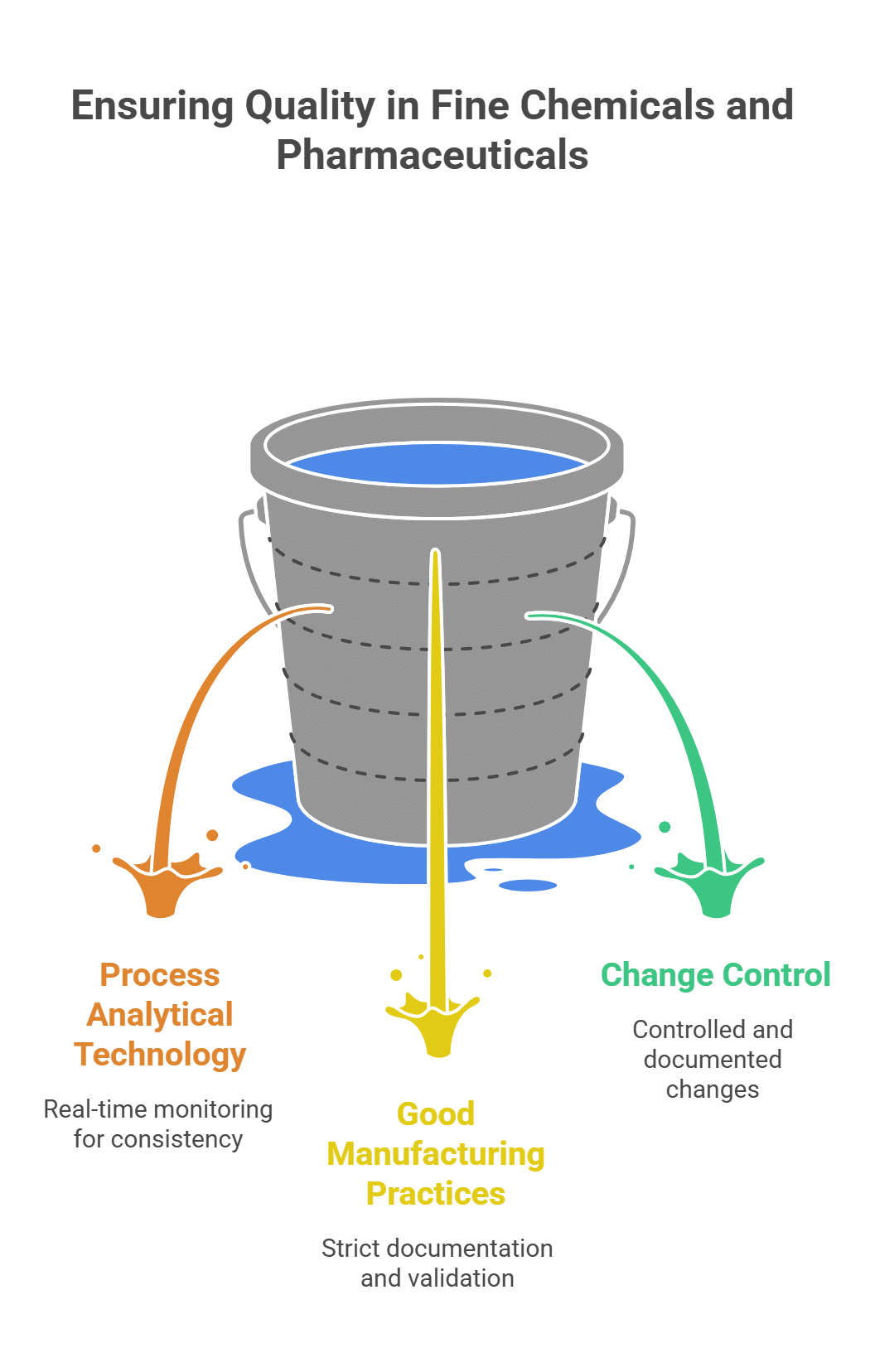
3. Unwavering Quality and Regulatory Control
In the fine chemical and pharmaceutical industries, quality is not negotiable. The regulatory burden increases exponentially as you move from R&D to commercial production. A process that is not designed with quality and compliance in mind from the outset will inevitably fail.
Key Challenges:
- Process Analytical Technology (PAT): Implementing real-time monitoring and control to ensure consistent product quality.
- Good Manufacturing Practices (GMP): Adhering to the strict documentation, validation, and control requirements of GMP regulations.
- Change Control: Managing any changes to the process, equipment, or raw materials in a controlled and documented manner.
Analysis of FDA enforcement data consistently shows that issues related to manufacturing processes and quality control are among the leading causes of drug recalls, highlighting the critical importance of getting the process right from the start [1].
Conclusion: The Foundation of Resilience
The art of scaling is the art of managing complexity. It is about taking a brilliant idea and translating it into a reliable, profitable, and safe industrial process. It requires a holistic approach that integrates chemistry, engineering, supply chain management, and quality assurance.
Ultimately, the ability to scale successfully is the foundation of a truly resilient business. A company that can consistently bring new innovations to market is a company that can adapt and thrive in a constantly changing world. But how do you build that resilience into the very fabric of your supply chain? That is the final piece of the puzzle, and the topic of our concluding article in this series.
References
[1] U.S. Food and Drug Administration (FDA). (2025). “Enforcement Reports.” https://www.fda.gov/safety/recalls-market-withdrawals-safety-alerts/enforcement-reports.
[2] American Institute of Chemical Engineers (AIChE). (2024). “The Challenges of Process Scale-Up.” https://www.aiche.org/cep/2024/04/challenges-process-scale.
[3] McKinsey & Company. (2025). “Scaling Digital and Analytics in Chemical Manufacturing.” https://www.mckinsey.com/industries/chemicals/our-insights/scaling-digital-and-analytics-in-chemical-manufacturing.