Unlocking the Value of Surplus Tetrachloroethylene in the Solvents Sector
Tetrachloroethylene Surplus Trading in the Solvents Sector | Efficient, Sustainable Chemical Inventory Solutions
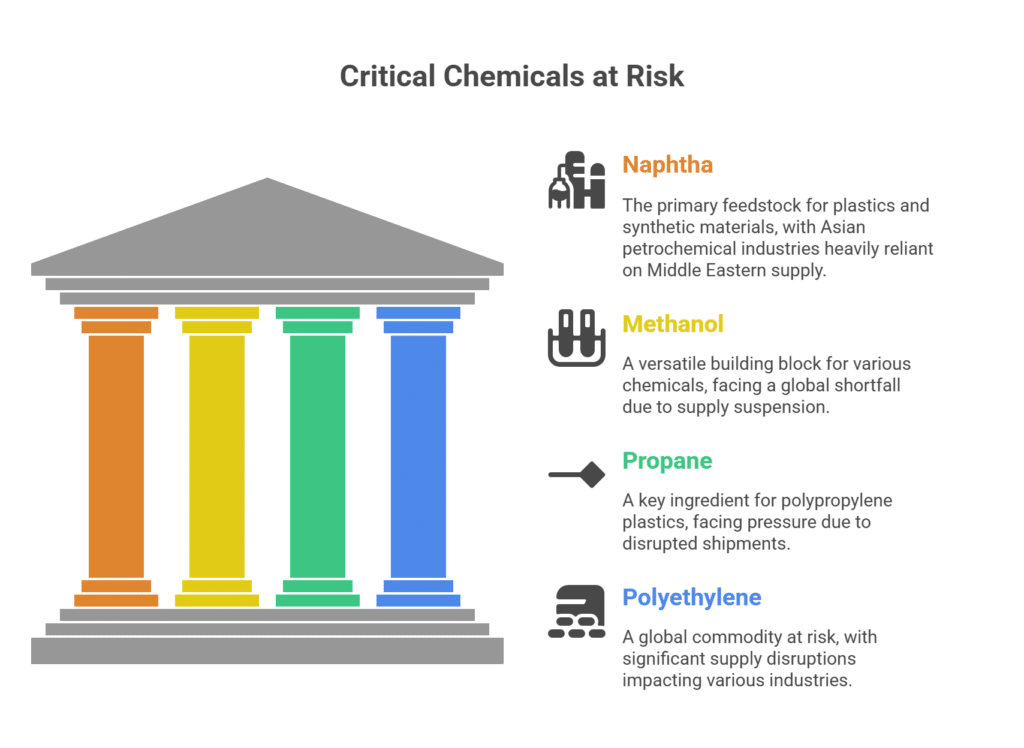
Tetrachloroethylene Applications in the Solvents Sector
Table of Contents
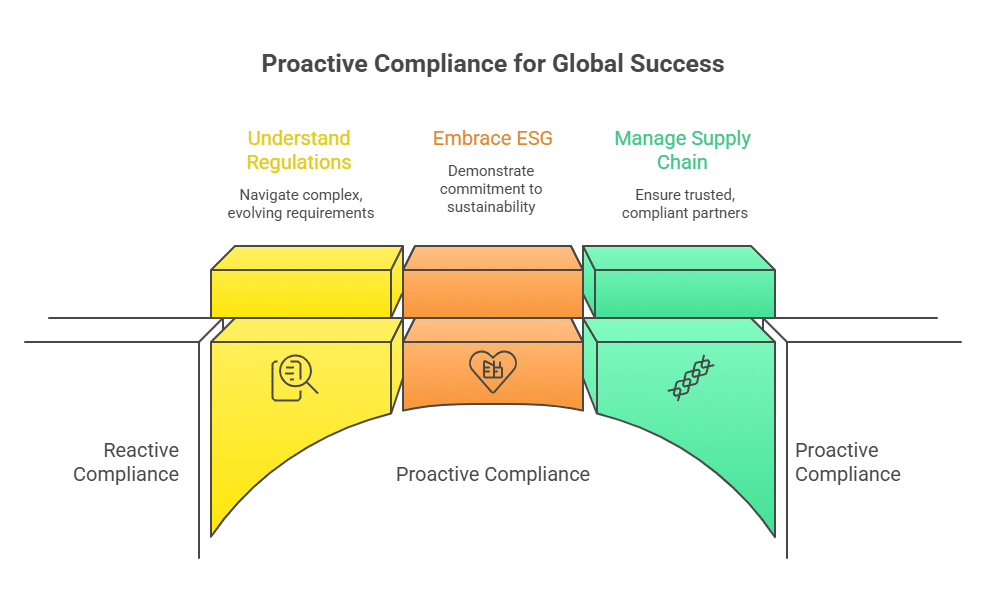
Successful Surplus Trading Boosts Efficiency in Industrial Cleaning
In a recent case study, a mid-sized manufacturing company was facing challenges with excess stocks of Tetrachloroethylene due to a change in their production process. By collaborating with a specialized surplus chemical trading platform, the company was able to turn their dormant inventory into an immediate revenue stream. They sold off a significant quantity of the solvent while simultaneously purchasing a high-quality, cost-effective batch for an emergency cleaning project. This transaction not only improved their cash flow and freed up storage space but also ensured they maintained uninterrupted production schedules. The initiative demonstrated how effective surplus trading can contribute to both operational efficiency and sustainable practices within the solvents sector.