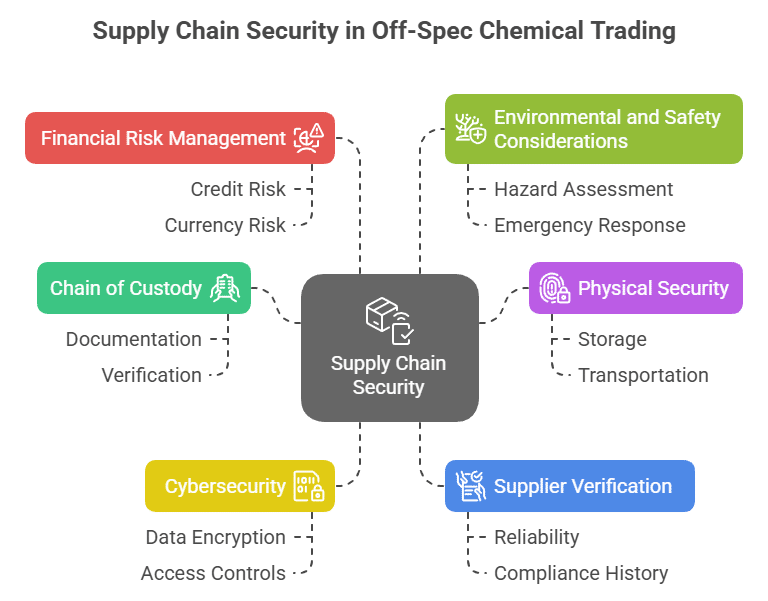
Sodium Hydroxide in Modern Paper & Pulp Manufacturing: Maximizing Value from Surplus Inventory
Sodium Hydroxide (caustic soda) is a highly versatile chemical integral to various industrial processes. In the chemical manufacturing and paper & pulp sector, it plays a crucial role in processes such as kraft pulping and pH adjustment. As surplus inventory, unused or excess stocks of this chemical can represent a hidden asset for companies when properly managed. With its well-known corrosive properties and robust performance in industrial applications, understanding its potential beyond immediate production needs opens up significant opportunities for both buyers and sellers.
Optimize Surplus Sodium Hydroxide Trading in Chemical Manufacturing & Paper & Pulp
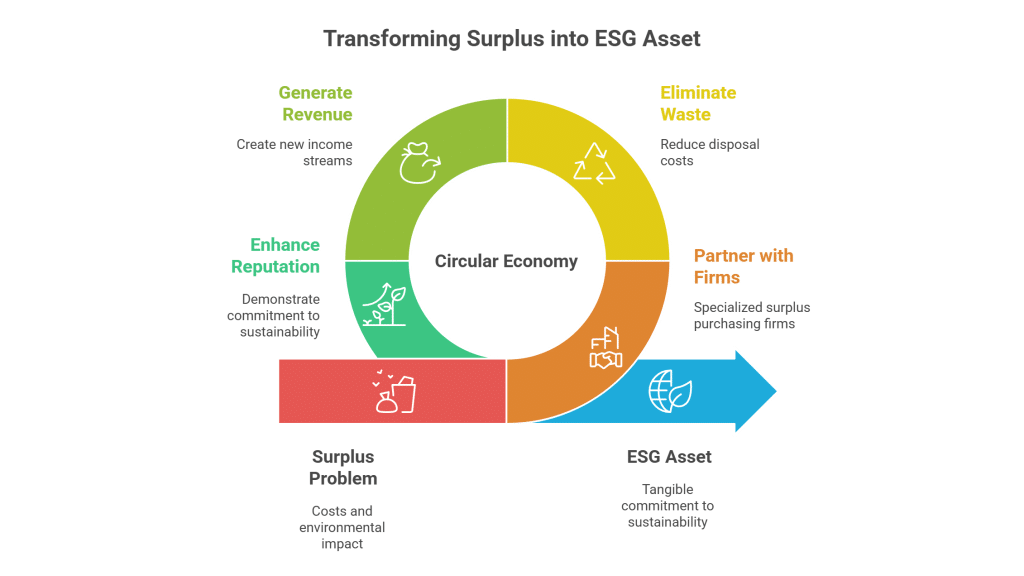
Trading surplus Sodium Hydroxide not only offers an effective approach to recoup costs, free up storage space, and avoid expensive disposal procedures but also contributes to environmental sustainability. Companies can profit by selling excess inventory which otherwise would incur regulatory and disposal burdens. Buyers gain cost savings and access reliable, high-quality materials essential for their operations. Embracing surplus trading practices promotes a circular economy, reducing waste and optimizing resource utilization—a win-win situation for both financial performance and environmental stewardship.
Sodium Hydroxide in Chemical Manufacturing & Paper & Pulp
For buyers, acquiring surplus Sodium Hydroxide offers significant cost savings on high-grade chemicals without compromising quality. It ensures a reliable supply chain, even in volatile market conditions, while supporting sustainable production practices. With access to well-stocked inventories, buyers can avoid production delays and reduce the costs associated with sourcing chemicals in emergency situations.
Sellers benefit by transforming aging or excess inventory into revenue, mitigating potential losses from chemical degradation, and reducing storage space requirements. Trading surplus also aids in compliance with hazardous waste regulations, leading to diminished disposal costs and lower environmental impact from excess chemicals. This approach helps companies maintain cleaner facilities and reallocate storage for more critical inventory items.
Table of Contents
Efficient Surplus Management: A Game-Changer for a Leading Paper Mill
A prominent paper mill recently leveraged its surplus Sodium Hydroxide inventory to streamline production and cut waste costs. By selling excess stocks that were stored for long periods, the facility not only freed up valuable storage space but also generated additional revenue. The cash flow was reinvested into advanced process controls, which resulted in improved pulp quality and reduced chemical waste. This strategic move resulted in enhanced sustainability credentials and a strengthened market position, illustrating the mutual benefits for both buyers and sellers in the surplus chemical marketplace.